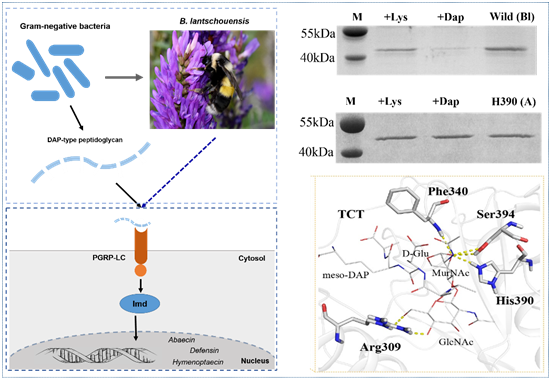
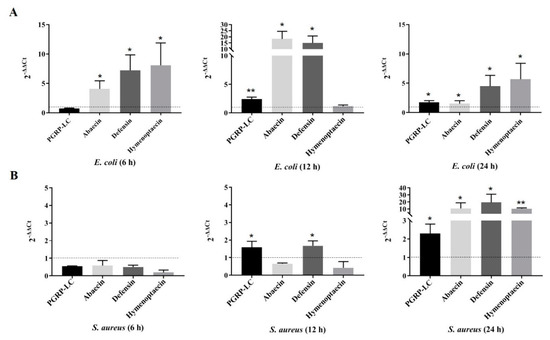
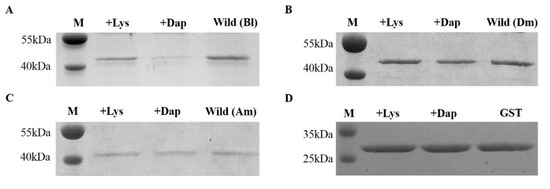
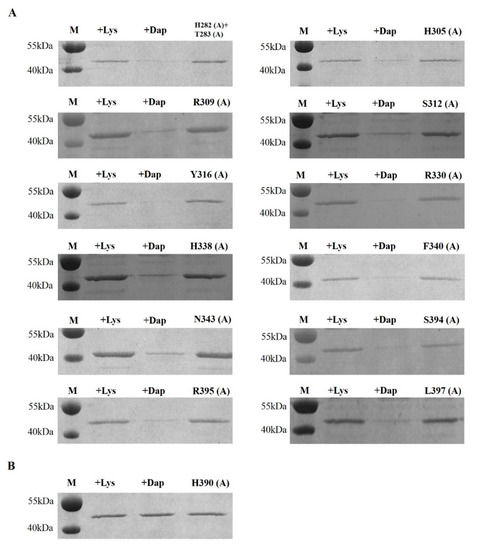
Peptidoglycan recognition proteins (PGRPs) play an important role in the defense against invading microbes via the recognition of the immunogenic substance peptidoglycan (PGN). Bees possess fewer PGRPs than Drosophila melanogaster and Anopheles gambiae but retain two important immune pathways, the Toll pathway and the Imd pathway, which can be triggered by the recognition of Dap-type PGN by PGRP-LCx with the assistance of PGRP-LCa in Drosophila. There are three isoforms of PGRP-LC including PGRP-LCx, PGRP-LCa and PGRP-LCy in Drosophila. Our previous study showed that a single PGRP-LC exists in bumblebees. In this present study, we prove that the bumblebee Bombus lantschouensis PGRP-LC (Bl-PGRP-LC) can respond to an infection with Gram-negative bacterium Escherichia coli through binding to the Dap-type PGNs directly, and that E. coli infection induces the quick and strong upregulation of PGRP-LC, abaecin and defensin. Moreover, the Bl-PGRP-LC exhibits a very strong affinity for the Dap-type PGN, much stronger than the affinity exhibited by the PGRP-LC from the more eusocial honeybee Apis mellifera (Am-PGRP-LC). In addition, mutagenesis experiments showed that the residue His390 is the anchor residue for the binding to the Dap-type PGN and forms a hydrogen bond with MurNAc rather than meso-Dap, which interacts with the anchor residue Arg413 of PGRP-LCx in Drosophila. Therefore, bumblebee PGRP-LC possesses exclusive characteristics for the immune response among insect PGRPs.
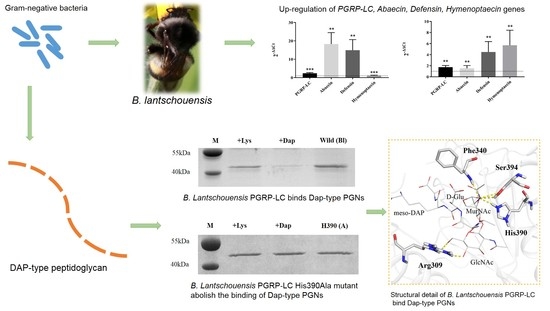
Graphical abstract
|